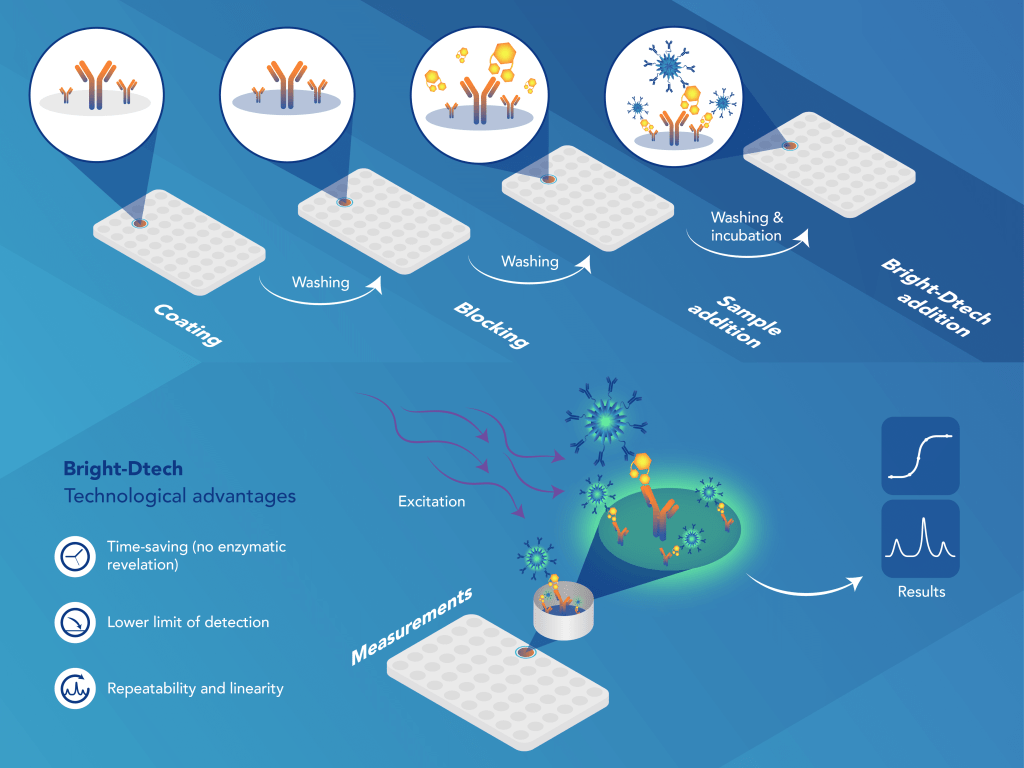
TR-FLISA Assay Principle
Fluorescence-linked immunosorbent assay (FLISA) is an analytical method which can be used for the detection and quantification of specific proteins, biomolecules, peptides, cytokines, hormones, antibodies, etc present in serum, plasma and cell culture supernatant.
Our Bright-Dtech™ technology brings a more sensible (high brightness resulting in an improved limit of detection) and stable (high photo-stability over days or weeks, resistance to photo-bleaching and durable signal intensity) approach.
In Bright-Dtech™ FLISA method, detection/secondary antibodies are conjugated to fluorescent nanoparticles, and a measure of the fluorescent intensity is detected by exposing the plate to light at a particular wavelength using time-resolved fluorescence reader, further increasing the sensitivity of the analysis.
Required material : Black microplates, plate shaker, Time-Resolved Fluorescence microplate reader.
Why using Bright-Dtech™ for FLISA applications?
Our technology brings benefits to users. Thanks to Bright-Dtech™ FLISA there is no substrate needed. We have a very low background, and an exceptional lifetime so no photobleaching. We have also a low LOD and LOQ. Finally, a low hands-on time.

Direct/Indirect FLISA
Direct/Indirect FLISA is used for the detection of antibodies against to an antigen immobilized to the surface of the multi-well plate. In indirect FLISA, a Bright-Dtech-conjugated secondary antibody against the host species of the primary antibody is used for detection. In direct FLISA, antibodies are directly conjugated to Bright-Dtech nanoparticles.
Example: Indirect FLISA assessing SARS-CoV-2 IgG antibody to nucleoprotein
Samples and standards are added to SARS-CoV-2 nucleoprotein-coated microwells plate. Unbound antibodies are washed away and Bright-Dtech-conjugated secondary antibody is added and incubated to form a complex. Time-resolved fluorescence is measured at a particular wavelength. The intensity of the resulting signal is directly proportional to the concentration of the antibody present in the sample.
We have shown that Bright-Dtech™ technology is able to significantly enhance the anti-SARS-CoV-2 IgG antibody detection thanks to the exceptional brightness of our fluorescent nanoparticles.

Figure 1. Indirect FLISA for anti-SARS-CoV-2 IgG antibody detection.
Bright-Dtech™ technology enhances the detection of anti-SARS-CoV-2 nucleoprotein IgG antibody due to its high specificity and sensitivity given by the time-resolved fluorescence signal. The assay range was 20-100 000 pg/mL leading to a very low/significant limit of detection around 30 pg/mL (using 3 standard deviation).

Sandwich-FLISA
Sandwich-FLISA assays are used to detect very low amounts of a specific antigen for a given sample. This method requires a couple of primary antibodies, each one recognizing a single epitope of the same antigen. The capture antibody is directly coated to the 96-well plate and binds the antigen present in the sample studied. The detection antibody is coupled to the Bright-Dtech™ fluorescent nanoparticles leading to an accurate and specific analyte quantification with a very low background. This last step may be performed with direct or indirect (requires a third antibody) sandwich detection.
Example: Sandwich-FLISA assay for SARS-CoV-2 nucleoprotein detection
This assay detects and quantifies human SARS-CoV-2 nucleoprotein in samples (serum, plasma or cell culture supernatant). Samples and standards are added to anti-SARS-CoV-2 nucleoprotein antibody-coated microwells plate leading to the antibody-antigen interaction. After incubation, several washes are performed to remove non-specific binding. Next, the Bright-Dtech™ conjugated detection antibody recognizes another epitope to complete the sandwich-FLISA. After excitation, the fluorescent signal emitted is directly linked to the nucleoprotein concentration.
We have shown that Bright-Dtech™ technology is able to significantly enhance the SARS-CoV-2 nucleoprotein detection thanks to the exceptional brightness of our fluorescent nanoparticles.
Our customized Bright-Dtech™ technology is available for all matched antibody pairs developed for classical sandwich ELISA allowing accurate detection and high signal/noise ratio for reliable results.

Figure 2. Sandwich FLISA for SARS-CoV-2 nucleoprotein detection.
Bright-Dtech™ technology enhances the detection of nucleoprotein due to its high specificity and sensitivity given by the time-resolved fluorescence signal. The assay range was 20-20 000 pg/mL leading to a very low/significant limit of detection around 18 pg/mL (using 3 standard deviation).